The 3D organization of the genome was shown recently to be intimately linked to its biological function and this opens an exciting new aspect of epigenetic regulation. Changes in nuclear architecture can affect cell fate and disruptions in genome topology can result in pathogenic phenotypes. Regulatory interactions with specific regions determine if genes will be turned on or off and thus are crucial to establish and maintain correct cell fate during development. Although essential, these interactions are difficult to study genome-wide because of the enormous complexity of possible contacts inside the nucleus where more than 2m of DNA are packed inside a tiny sphere (the cell nucleus) of ~10mm diameter.
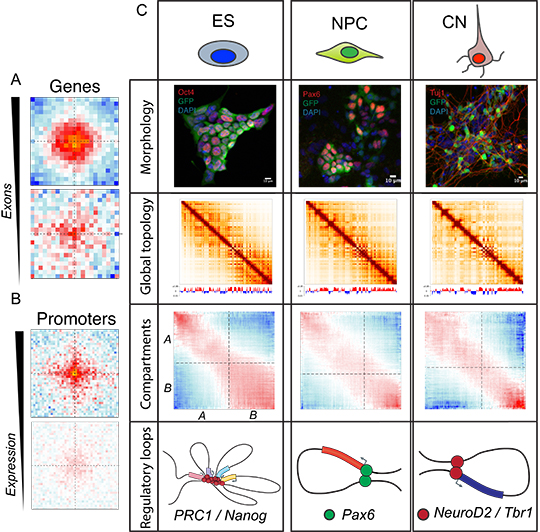
Now, using state-of-the-art methods to purify specific cell populations in vivo and map their 3D nuclear architecture at an unprecedented resolution, a team of researchers based at the Institute of Human Genetics of the CNRS and the University of Montpellier has shown that the changes in genome topology occur across multiple scales during mouse brain development. The researchers showed that when genes are switched on they are usually associated with insulation, thus preventing contacts from crossing them. However, artificially turning genes on did not lead to the creation of new chromatin boundaries, suggesting that chromatin organization may precede gene expression and may depend of factors that remain to be identified. Furthermore, the researchers discovered previously unknown associations between splicing and 3D genome organization, whereby heavily spliced genes preferentially contact each other inside the nucleus.
In addition to these findings, which were valid for both pluripotent and differentiated cells, they discovered that key regulatory interactions are highly dynamic and cell-type specific. A strong Polycomb-linked contacts network in stem cells is disrupted during neural differentiation, while chromatin interactions between neuronal transcription factors are established. These contacts were specific to regions bound by the same transcription factor, pointing to a potential novel role to regulate cell fate in the brain. Regulatory contacts between an enhancer region and the gene are dynamically regulated and are strengthened only when the gene is switched on.
These findings illustrate how 3D nuclear architecture is deeply linked to the normal physiological and pathological function of the brain in vivo. They have implications for several brain-related diseases such as intellectual disability and autism, which are frequently associated with chromatin-remodeling complexes.
Further information
http://www.cnrs.fr/insb/recherche/parutions/articles2017/g-cavalli-EN.html
Multi-scale 3D genome rewiring during mouse neural development.
Bonev, B., Mendelson Cohen, N., Szabo, Q., Fritsch, L., Papadopoulos, G., Lubling, Y., Xu, X., Lv, X., Hugnot, J.-P., Tanay, A, and Cavalli, G.
Cell Volume 171, Issue 3, p557–572.e24, 19 October 2017DOI: http://dx.doi.org/10.1016/j.cell.2017.09.043
Research contacts
Giacomo Cavalli
Institut de Génétique Humaine
CNRS UMR 9002 – Université de Montpellier
141, rue de la Cardonille
34396 Montpellier – Cedex 5
Boyan Bonev
Institut de Génétique Humaine
CNRS UMR 9002 – Université de Montpellier
141, rue de la Cardonille
34396 Montpellier – Cedex 5

